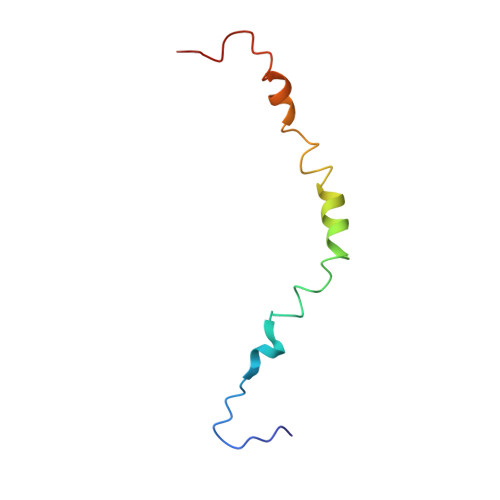
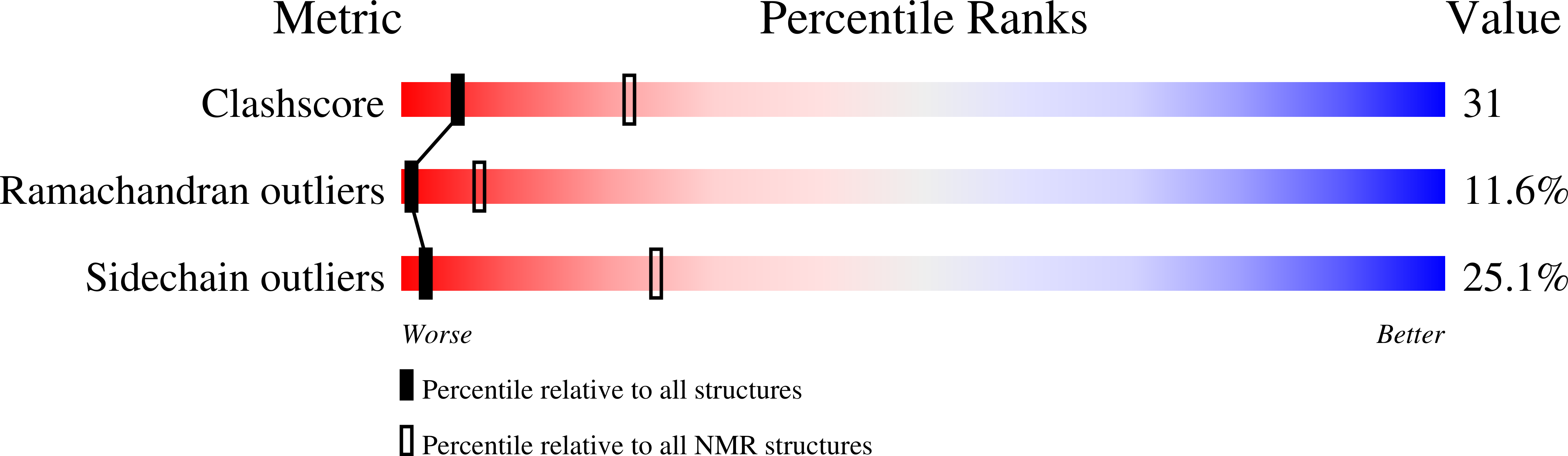Structural features of the apelin receptor N-terminal tail and first transmembrane segment implicated in ligand binding and receptor trafficking.
Langelaan, D.N., Reddy, T., Banks, A.W., Dellaire, G., Dupre, D.J., Rainey, J.K.(2013) Biochim Biophys Acta 1828: 1471-1483
- PubMed: 23438363
- DOI: https://doi.org/10.1016/j.bbamem.2013.02.005
- Primary Citation of Related Structures:
2LOU - PubMed Abstract:
G-protein coupled receptors (GPCRs) comprise a large family of membrane proteins with rich functional diversity. Signaling through the apelin receptor (AR or APJ) influences the cardiovascular system, central nervous system and glucose regulation. Pathophysiological involvement of apelin has been shown in atherosclerosis, cancer, human immunodeficiency virus-1 (HIV-1) infection and obesity. Here, we present the high-resolution nuclear magnetic resonance (NMR) spectroscopy-based structure of the N-terminus and first transmembrane (TM) segment of AR (residues 1-55, AR55) in dodecylphosphocholine micelles. AR55 consists of two disrupted helices, spanning residues D14-K25 and A29-R55(1.59). Molecular dynamics (MD) simulations of AR built from a hybrid of experimental NMR and homology model-based restraints allowed validation of the AR55 structure in the context of the full-length receptor in a hydrated bilayer. AR55 structural features were functionally probed using mutagenesis in full-length AR through monitoring of apelin-induced extracellular signal-regulated kinase (ERK) phosphorylation in transiently transfected human embryonic kidney (HEK) 293A cells. Residues E20 and D23 form an extracellular anionic face and interact with lipid headgroups during MD simulations in the absence of ligand, producing an ideal binding site for a cationic apelin ligand proximal to the membrane-water interface, lending credence to membrane-catalyzed apelin-AR binding. In the TM region of AR55, N46(1.50) is central to a disruption in helical character. G42(1.46), G45(1.49) and N46(1.50), which are all involved in the TM helical disruption, are essential for proper trafficking of AR. In summary, we introduce a new correlative NMR spectroscopy and computational biochemistry methodology and demonstrate its utility in providing some of the first high-resolution structural information for a peptide-activated GPCR TM domain.
Organizational Affiliation:
Department of Biochemistry & Molecular Biology, Dalhousie University, Halifax, Nova Scotia, Canada.