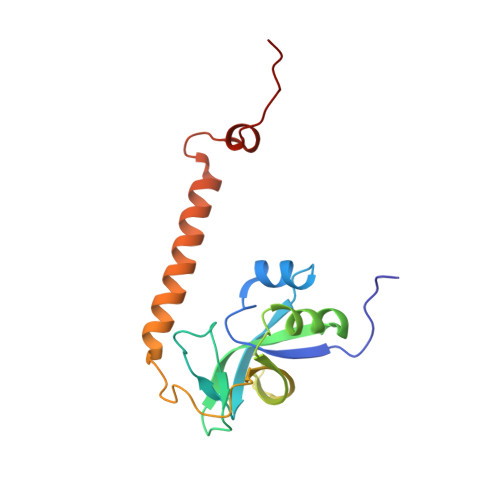
Solution structure of At3g28950 from Arabidopsis thaliana.
de la Cruz, N.B., Peterson, F.C., Volkman, B.F.(2008) Proteins 71: 546-551
- PubMed: 18214976
- DOI: https://doi.org/10.1002/prot.21936
- Primary Citation of Related Structures:
2JQV - PubMed Abstract:
We determined the solution structure of At3g28950 from A. thaliana, a homolog of At5g39720, whose structure we solved earlier. The secondary structure of the 165-aa protein consists of a 5-strand antiparallel beta-barrel domain flanked by two alpha-helices and a 2-strand beta-sheet; an additional free C-terminal alpha-helix extends into solution. Bioinformatic searches and analyses suggest that members of this growing set of structurally related proteins have been recruited to serve a wide variety of functions ranging from gamma-glutamyl cyclotransferase activity to participation in plant responses to chemical and biotic stimuli. Expression of a human homolog is elevated in bladder cancer tissues. Expression patterns for At3g28950 and its Arabidopsis paralogs suggest that each one evolved a different physiological role. The At3g28950 structure was solved as part of a structural genomics effort, and the results demonstrate how such a project can further understanding of genome evolution in addition to sequence-structure and structure-function relationships. Proteins 2008. (c) 2008 Wiley-Liss, Inc.
Organizational Affiliation:
Department of Biochemistry and Center for Eukaryotic Structural Genomics, Medical College of Wisconsin, Milwaukee, Wisconsin 53226, USA.