Crystal structure of the N-acetylglucosamine-6-phosphate deacetylase from Escherichia Coli
Fedorov, A.A., Fedorov, E.V., Xiang, D.F., Raushel, F.M., Almo, S.C.To be published.
Experimental Data Snapshot
wwPDB Validation 3D Report Full Report
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| N-acetylglucosamine-6-phosphate deacetylase | 382 | Escherichia coli K-12 | Mutation(s): 0 Gene Names: nagA EC: 3.5.1.25 | 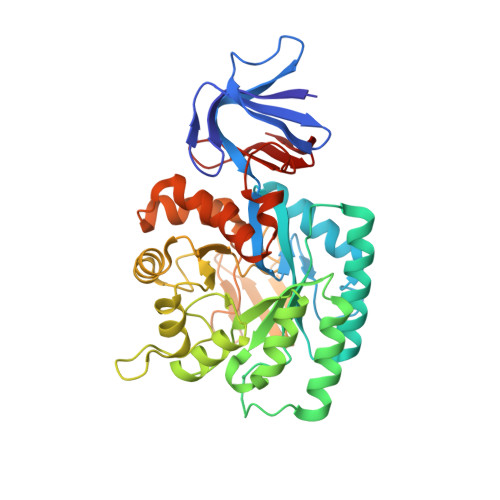 | |
UniProt | |||||
Find proteins for P0AF18 (Escherichia coli (strain K12)) Explore P0AF18 Go to UniProtKB: P0AF18 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P0AF18 | ||||
Sequence AnnotationsExpand | |||||
| |||||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 82.226 | α = 90 |
| b = 114.023 | β = 90 |
| c = 80.215 | γ = 90 |
| Software Name | Purpose |
|---|---|
| HKL-2000 | data collection |
| SCALEPACK | data scaling |
| SOLVE | phasing |
| CNS | refinement |
| HKL-2000 | data reduction |