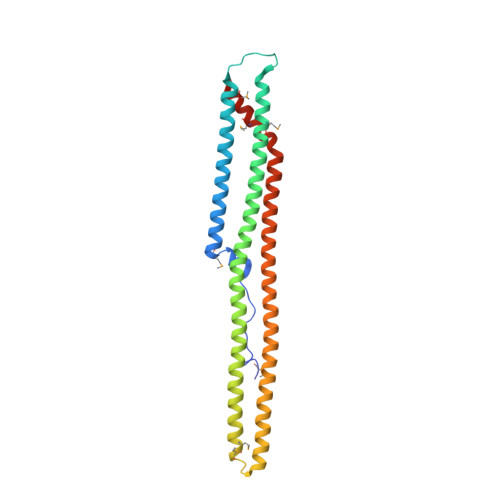
A unique fold of phospholipase C-beta mediates dimerization and interaction with G alpha q.
Singer, A.U., Waldo, G.L., Harden, T.K., Sondek, J.(2002) Nat Struct Biol 9: 32-36
- PubMed: 11753430
- DOI: https://doi.org/10.1038/nsb731
- Primary Citation of Related Structures:
1JAD - PubMed Abstract:
GTP-bound subunits of the Gq family of G alpha subunits directly activate phospholipase C-beta (PLC-beta) isozymes to produce the second messengers inositol 1,4,5-trisphosphate and diacylglycerol. PLC-betas are GTPase activating proteins (GAPs) that also promote the formation of GDP-bound, inactive G beta subunits. Both phospholipase activation by G alpha-GTP subunits and GAP activity require a C-terminal region unique to PLC-beta isozymes. The crystal structure of the C-terminal region from an avian PLC-beta, determined at 2.4 A resolution, reveals a novel fold composed almost entirely of three long helices forming a coiled-coil that dimerizes along its long axis in an antiparallel orientation. The dimer interface is extensive ( approximately 3,200 A(2)), and, based on gel exclusion chromatography, full length PLC-betas are dimeric, indicating that PLC-betas likely function as dimers. Sequence conservation, mutational data and molecular modeling show that an electrostatically positive surface of the dimer contains the major determinants for binding G beta q. Effector dimerization, as highlighted by PLC-betas, provides a viable mechanism for regulating signaling cascades linked to heterotrimeric G proteins.
Organizational Affiliation:
Department of Pharmacology, The University of North Carolina at Chapel Hill, North Carolina 27599, USA.