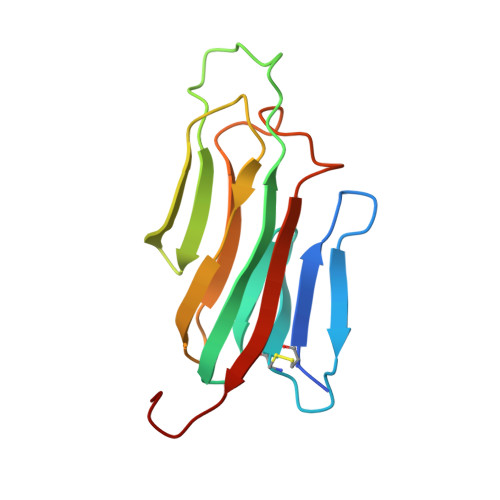
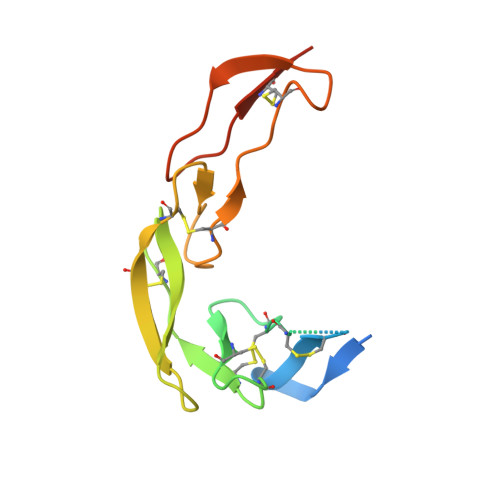
Structures of mouse and human GITR-GITRL complexes reveal unique TNF superfamily interactions.
Wang, F., Chau, B., West, S.M., Kimberlin, C.R., Cao, F., Schwarz, F., Aguilar, B., Han, M., Morishige, W., Bee, C., Dollinger, G., Rajpal, A., Strop, P.(2021) Nat Commun 12: 1378-1378
- PubMed: 33654081
- DOI: https://doi.org/10.1038/s41467-021-21563-z
- Primary Citation of Related Structures:
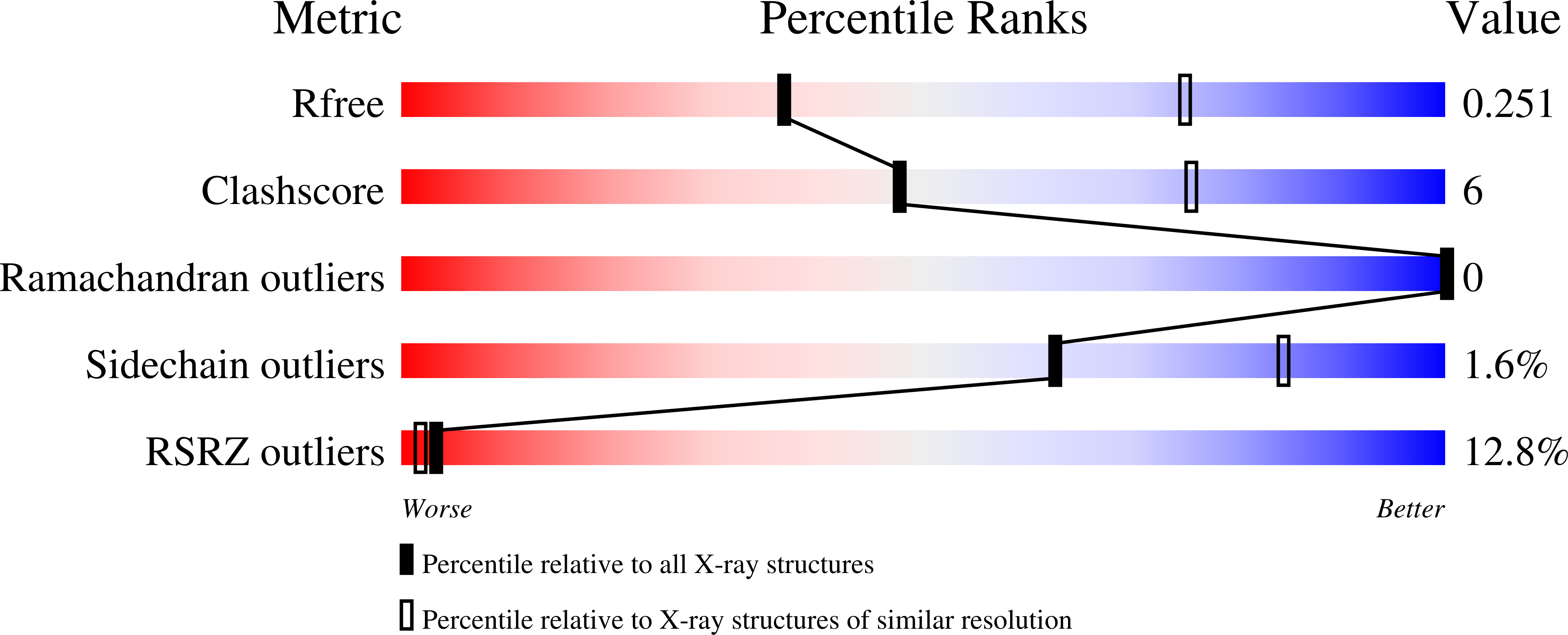
7KHD, 7KHX - PubMed Abstract:
Glucocorticoid-induced tumor necrosis factor receptor-related protein (GITR) and GITR ligand (GITRL) are members of the tumor necrosis superfamily that play a role in immune cell signaling, activation, and survival. GITR is a therapeutic target for directly activating effector CD4 and CD8 T cells, or depleting GITR-expressing regulatory T cells (Tregs), thereby promoting anti-tumor immune responses. GITR activation through its native ligand is important for understanding immune signaling, but GITR structure has not been reported. Here we present structures of human and mouse GITR receptors bound to their cognate ligands. Both species share a receptor-ligand interface and receptor-receptor interface; the unique C-terminal receptor-receptor enables higher order structures on the membrane. Human GITR-GITRL has potential to form a hexameric network of membrane complexes, while murine GITR-GITRL complex forms a linear chain due to dimeric interactions. Mutations at the receptor-receptor interface in human GITR reduce cell signaling with in vitro ligand binding assays and minimize higher order membrane structures when bound by fluorescently labeled ligand in cell imaging experiments.
Organizational Affiliation:
Discovery Biotherapeutics, Bristol Myers Squibb, Redwood City, CA, USA.