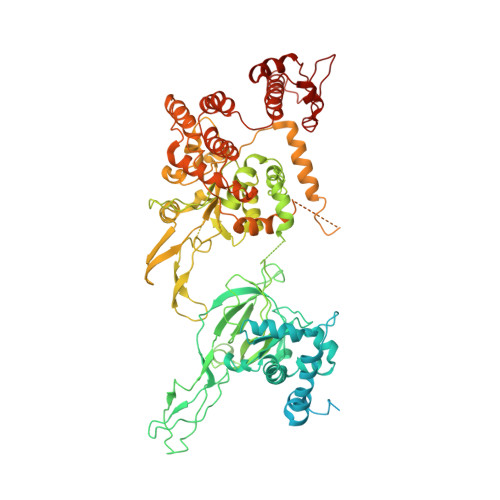
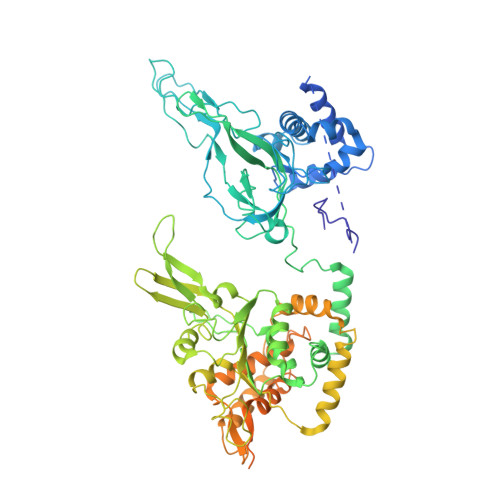
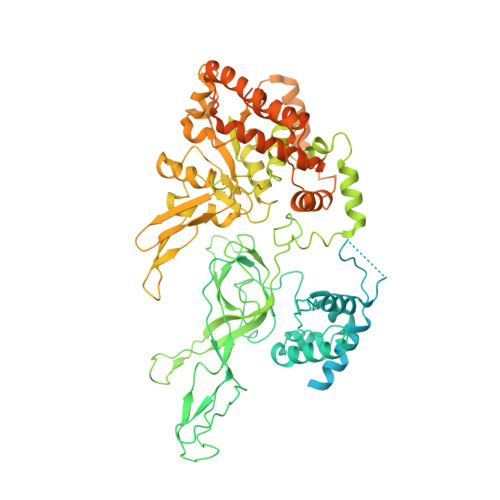
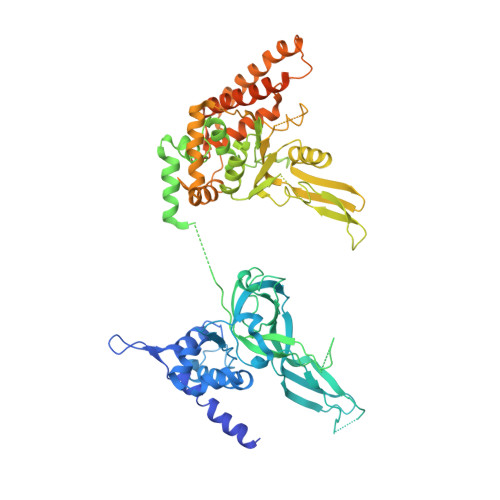
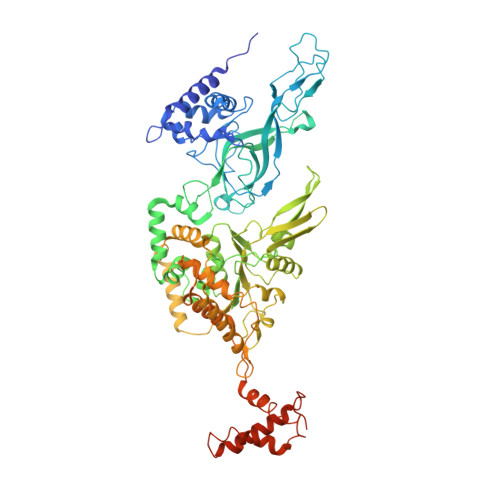
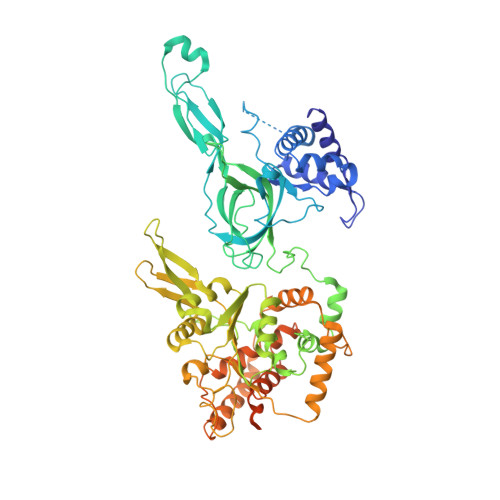
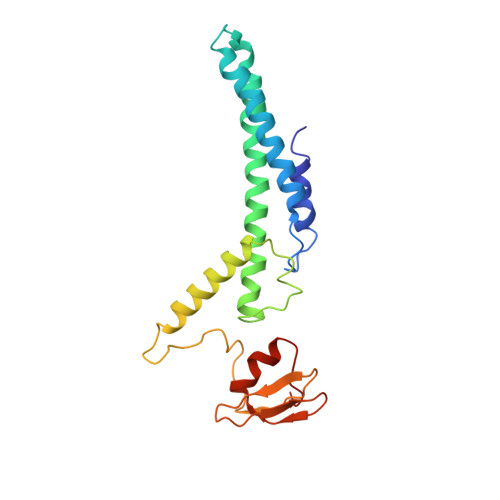
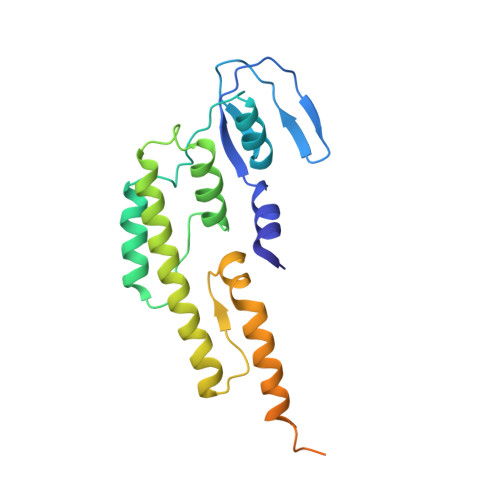
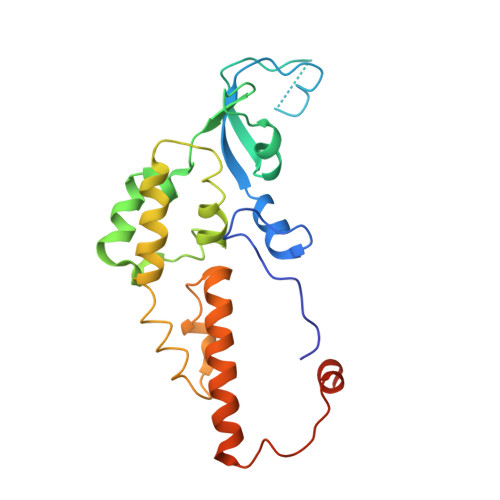
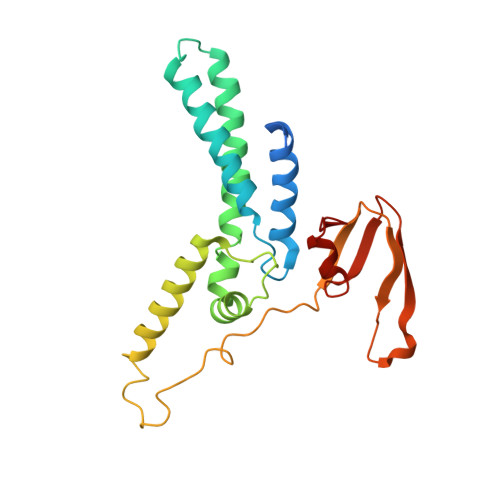
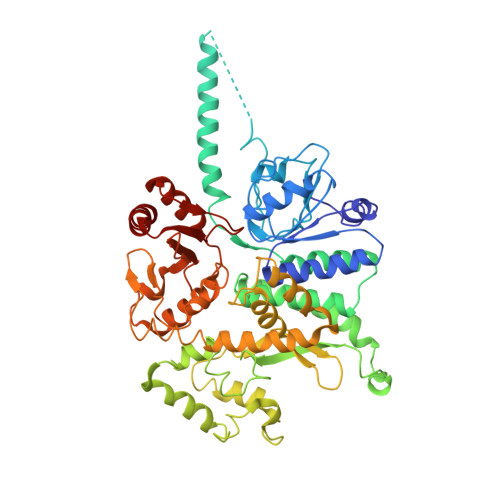
CryoEM structures of human CMG-ATP gamma S-DNA and CMG-AND-1 complexes.
Rzechorzek, N.J., Hardwick, S.W., Jatikusumo, V.A., Chirgadze, D.Y., Pellegrini, L.(2020) Nucleic Acids Res 48: 6980-6995
- PubMed: 32453425
- DOI: https://doi.org/10.1093/nar/gkaa429
- Primary Citation of Related Structures:
6XTX, 6XTY - PubMed Abstract:
DNA unwinding in eukaryotic replication is performed by the Cdc45-MCM-GINS (CMG) helicase. Although the CMG architecture has been elucidated, its mechanism of DNA unwinding and replisome interactions remain poorly understood. Here we report the cryoEM structure at 3.3 Å of human CMG bound to fork DNA and the ATP-analogue ATPγS. Eleven nucleotides of single-stranded (ss) DNA are bound within the C-tier of MCM2-7 AAA+ ATPase domains. All MCM subunits contact DNA, from MCM2 at the 5'-end to MCM5 at the 3'-end of the DNA spiral, but only MCM6, 4, 7 and 3 make a full set of interactions. DNA binding correlates with nucleotide occupancy: five MCM subunits are bound to either ATPγS or ADP, whereas the apo MCM2-5 interface remains open. We further report the cryoEM structure of human CMG bound to the replisome hub AND-1 (CMGA). The AND-1 trimer uses one β-propeller domain of its trimerisation region to dock onto the side of the helicase assembly formed by Cdc45 and GINS. In the resulting CMGA architecture, the AND-1 trimer is closely positioned to the fork DNA while its CIP (Ctf4-interacting peptide)-binding helical domains remain available to recruit partner proteins.
Organizational Affiliation:
Department of Biochemistry, Tennis Court Road, Cambridge CB2 1GA, UK.