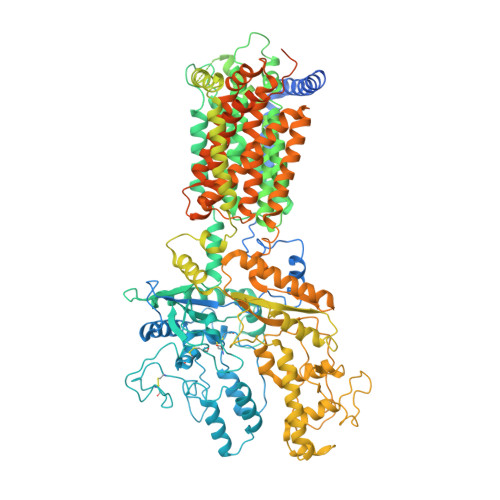
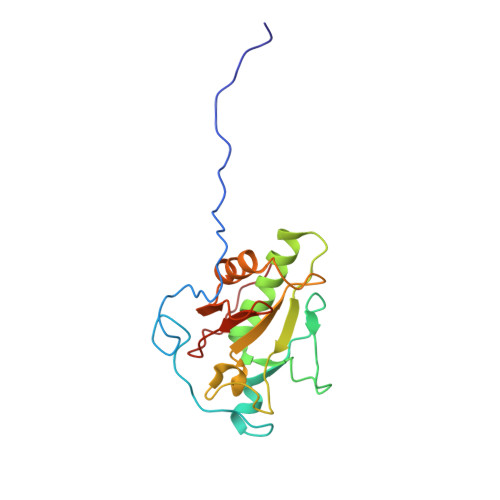
Inhibition of tetrameric Patched1 by Sonic Hedgehog through an asymmetric paradigm.
Qian, H., Cao, P., Hu, M., Gao, S., Yan, N., Gong, X.(2019) Nat Commun 10: 2320-2320
- PubMed: 31127104
- DOI: https://doi.org/10.1038/s41467-019-10234-9
- Primary Citation of Related Structures:
6N7G, 6N7H, 6N7K - PubMed Abstract:
The Hedgehog (Hh) pathway controls embryonic development and postnatal tissue maintenance and regeneration. Inhibition of Hh receptor Patched (Ptch) by the Hh ligands relieves suppression of signaling cascades. Here, we report the cryo-EM structure of tetrameric Ptch1 in complex with the palmitoylated N-terminal signaling domain of human Sonic hedgehog (ShhN p ) at a 4:2 stoichiometric ratio. The structure shows that four Ptch1 protomers are organized as a loose dimer of dimers. Each dimer binds to one ShhN p through two distinct inhibitory interfaces, one mainly through the N-terminal peptide and the palmitoyl moiety of ShhN p and the other through the Ca 2+ -mediated interface on ShhN p . Map comparison reveals that the cholesteryl moiety of native ShhN occupies a recently identified extracellular steroid binding pocket in Ptch1. Our structure elucidates the tetrameric assembly of Ptch1 and suggests an asymmetric mode of action of the Hh ligands for inhibiting the potential cholesterol transport activity of Ptch1.
Organizational Affiliation:
Department of Molecular Biology, Princeton University, Princeton, NJ, 08544, USA.