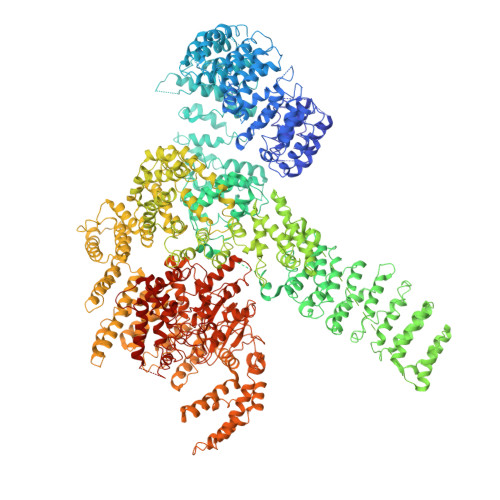
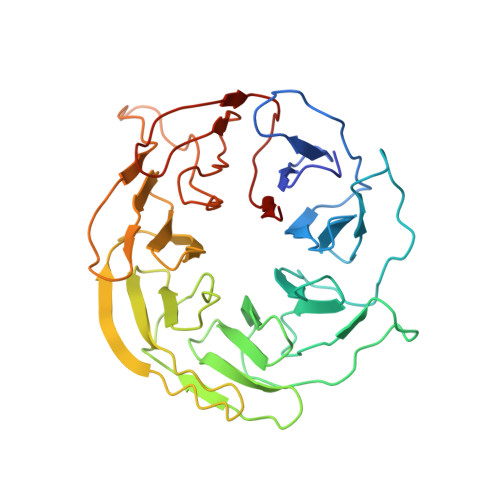
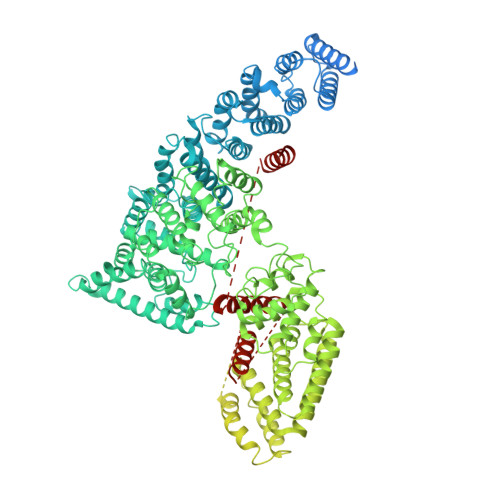
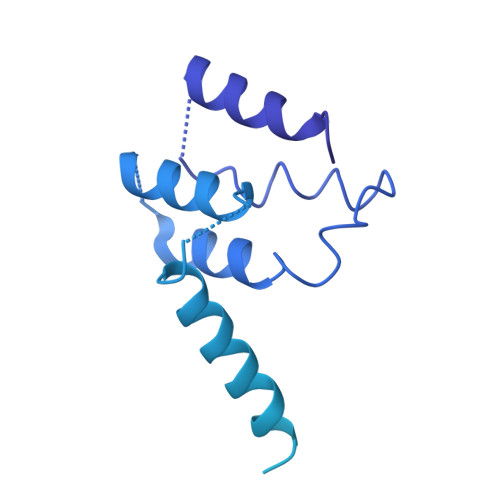
Cryo-EM structure of human mTOR complex 2.
Chen, X., Liu, M., Tian, Y., Li, J., Qi, Y., Zhao, D., Wu, Z., Huang, M., Wong, C.C.L., Wang, H.W., Wang, J., Yang, H., Xu, Y.(2018) Cell Res 28: 518-528
- PubMed: 29567957
- DOI: https://doi.org/10.1038/s41422-018-0029-3
- Primary Citation of Related Structures:
5ZCS - PubMed Abstract:
Mechanistic target of rapamycin (mTOR) complex 2 (mTORC2) plays an essential role in regulating cell proliferation through phosphorylating AGC protein kinase family members, including AKT, PKC and SGK1. The functional core complex consists of mTOR, mLST8, and two mTORC2-specific components, Rictor and mSin1. Here we investigated the intermolecular interactions within mTORC2 complex and determined its cryo-electron microscopy structure at 4.9 Å resolution. The structure reveals a hollow rhombohedral fold with a 2-fold symmetry. The dimerized mTOR serves as a scaffold for the complex assembly. The N-terminal half of Rictor is composed of helical repeat clusters and binds to mTOR through multiple contacts. mSin1 is located close to the FRB domain and catalytic cavity of mTOR. Rictor and mSin1 together generate steric hindrance to inhibit binding of FKBP12-rapamycin to mTOR, revealing the mechanism for rapamycin insensitivity of mTORC2. The mTOR dimer in mTORC2 shows more compact conformation than that of mTORC1 (rapamycin sensitive), which might result from the interaction between mTOR and Rictor-mSin1. Structural comparison shows that binding of Rictor and Raptor (mTORC1-specific component) to mTOR is mutually exclusive. Our study provides a basis for understanding the assembly of mTORC2 and a framework to further characterize the regulatory mechanism of mTORC2 pathway.
Organizational Affiliation:
Fudan University Shanghai Cancer Center, Institutes of Biomedical Sciences, Shanghai Medical College of Fudan University, Shanghai, 200032, China.