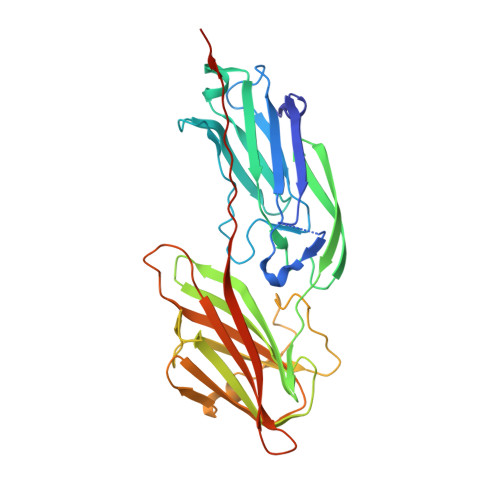
Staphylococcus aureus SdrE captures complement factor H's C-terminus via a novel 'close, dock, lock and latch' mechanism for complement evasion
Zhang, Y., Wu, M., Hang, T., Wang, C., Yang, Y., Pan, W., Zang, J., Zhang, M., Zhang, X.(2017) Biochem J 474: 1619-1631
- PubMed: 28258151
- DOI: https://doi.org/10.1042/BCJ20170085
- Primary Citation of Related Structures:
5WTA, 5WTB - PubMed Abstract:
Complement factor H (CFH) is a soluble complement regulatory protein essential for the down-regulation of the alternative pathway on interaction with specific markers on the host cell surface. It recognizes the complement component 3b (C3b) and 3d (C3d) fragments in addition to self cell markers (i.e. glycosaminoglycans, sialic acid) to distinguish host cells that deserve protection from pathogens that should be eliminated. The Staphylococcus aureus surface protein serine-aspartate repeat protein E (SdrE) was previously reported to bind human CFH as an immune-evasion tactic. However, the molecular mechanism underlying SdrE-CFH-mediated immune evasion remains unknown. In the present study, we identified a novel region at CFH's C-terminus (CFH 1206-1226 ), which binds SdrE N2 and N3 domains (SdrE N2N3 ) with high affinity, and determined the crystal structures of apo-SdrE N2N3 and the SdrE N2N3 -CFH 1206-1226 complex. Comparison of the structure of the CFH-SdrE complex with other CFH structures reveals that CFH's C-terminal tail flips from the main body to insert into the ligand-binding groove of SdrE. In addition, SdrE N2N3 adopts a 'close' state in the absence of CFH, which undergoes a large conformational change on CFH binding, suggesting a novel 'close, dock, lock and latch' (CDLL) mechanism for SdrE to recognize its ligand. Our findings imply that SdrE functions as a 'clamp' to capture CFH's C-terminal tail via a unique CDLL mechanism and sequesters CFH on the surface of S. aureus for complement evasion.
Organizational Affiliation:
School of Life Science, Anhui University, 111 Jiulong Road, Hefei 230601, China.