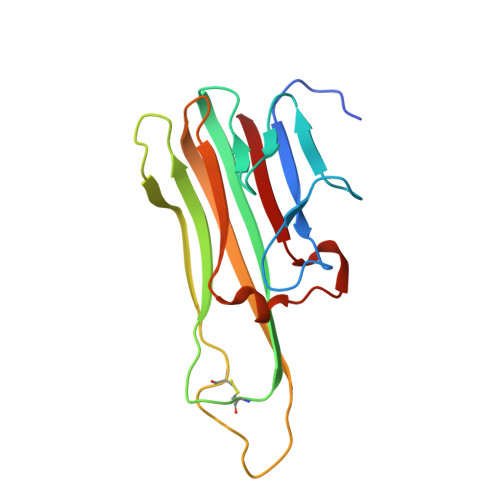
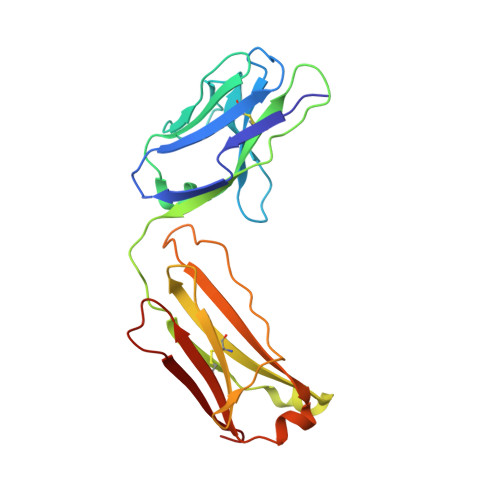
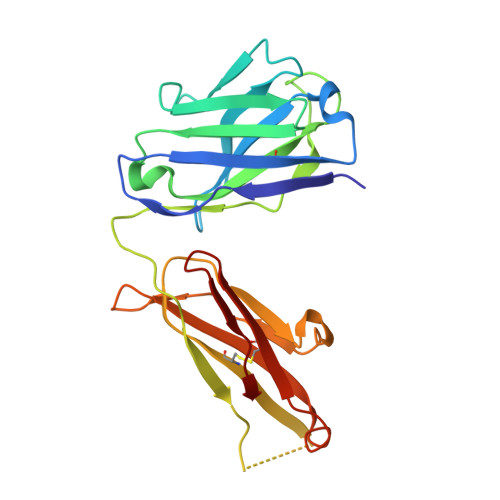
Comparison of the inhibition mechanisms of adalimumab and infliximab in treating tumor necrosis factor alpha-associated diseases from a molecular view
Hu, S., Liang, S.Y., Guo, H., Zhang, D., Li, H., Wang, X., Yang, W., Qian, W., Hou, S., Wang, H., Guo, Y.J., Lou, Z.Y.(2013) J Biol Chem 288: 27059-27067
- PubMed: 23943614
- DOI: https://doi.org/10.1074/jbc.M113.491530
- Primary Citation of Related Structures:
3WD5 - PubMed Abstract:
TNFα-targeting therapy with the use of the drugs Etanercept, Infliximab, and Adalimumab is used in the clinical treatment of various inflammatory and immune diseases. Although all of these reagents function to disrupt the interaction between TNFα and its receptors, clinical investigations showed the advantages of Adalimumab treatment compared with Etanercept and Infliximab. However, the underlying molecular mechanism of action of Adalimumab remains unclear. In our previous work, we presented structural data on how Infliximab binds with the E-F loop of TNFα and functions as a TNFα receptor-binding blocker. To further elucidate the variations between TNFα inhibitors, we solved the crystal structure of TNFα in complex with Adalimumab Fab. The structural observation and the mutagenesis analysis provided direct evidence for identifying the Adalimumab epitope on TNFα and revealed the mechanism of Adalimumab inhibition of TNFα by occupying the TNFα receptor-binding site. The larger antigen-antibody interface in TNFα Adalimumab also provided information at a molecular level for further understanding the clinical advantages of Adalimumab therapy compared with Infliximab.
Organizational Affiliation:
Laboratory of Structural Biology and Ministry of Education Laboratory of Protein Science, School of Medicine, Tsinghua University, Beijing 100084, China; International Joint Cancer Institute, Second Military Medical University, Shanghai 200433, China.