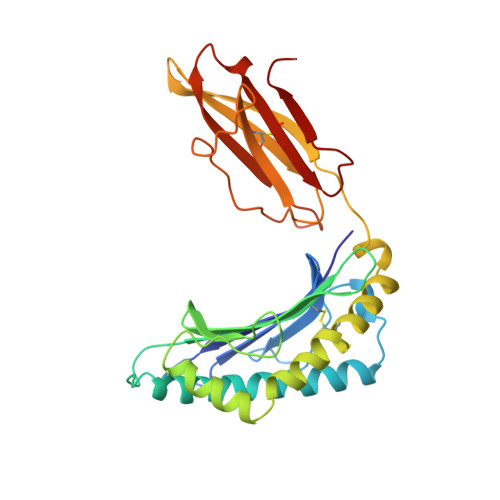
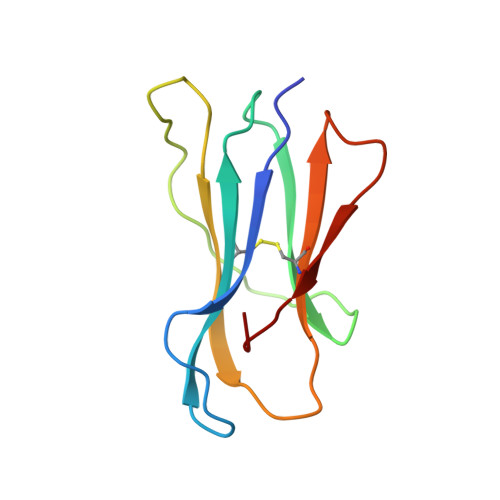
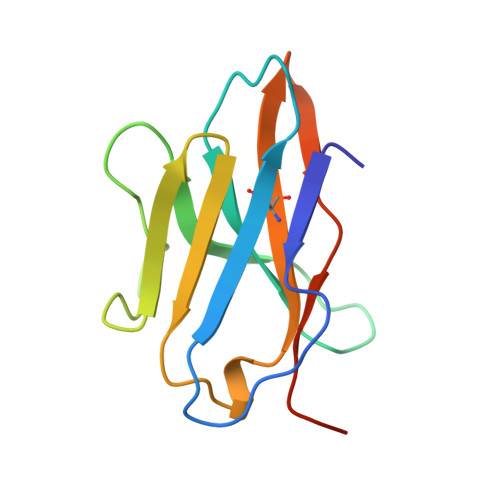
Crystal structure of the complex between human CD8alpha(alpha) and HLA-A2.
Gao, G.F., Tormo, J., Gerth, U.C., Wyer, J.R., McMichael, A.J., Stuart, D.I., Bell, J.I., Jones, E.Y., Jakobsen, B.K.(1997) Nature 387: 630-634
- PubMed: 9177355
- DOI: https://doi.org/10.1038/42523
- Primary Citation of Related Structures:
1AKJ - PubMed Abstract:
The dimeric cell-surface glycoprotein CD8 is crucial to the positive selection of cytotoxic T cells in the thymus. The homodimer CD8alpha(alpha) or the heterodimer alpha beta stabilizes the interaction of the T-cell antigen receptor (TCR) with major histocompatibility complex (MHC) class I/peptide by binding to the class I molecule. Here we report the crystal structure at 2.7 A resolution of a complex between CD8alpha(alpha) and the human MHC molecule HLA-A2, which is associated with peptide. CD8alpha(alpha) binds one HLA-A2/peptide molecule, interfacing with the alpha2 and alpha3 domains of HLA-A2 and also contacting beta2-microglobulin. A flexible loop of the alpha3 domain (residues 223-229) is clamped between the complementarity-determining region (CDR)-like loops of the two CD8 subunits in the classic manner of an antibody-antigen interaction, precluding the binding of a second MHC molecule. The position of the alpha3 domain is different from that in uncomplexed HLA-A2, being most similar to that in the TCR/Tax/HLA-A2 complex, but no conformational change extends to the MHC/peptide surface presented for TCR recognition. Although these shifts in alpha3 may provide a synergistic modulation of affinity, the binding of CD8 to MHC is clearly consistent with an avidity-based contribution from CD8 to TCR-peptide-MHC interactions.
Organizational Affiliation:
Molecular Immunology Group, Nuffield Department of Clinical Medicine, Institute of Molecular Medicine, John Radcliffe Hospital, Oxford, UK.