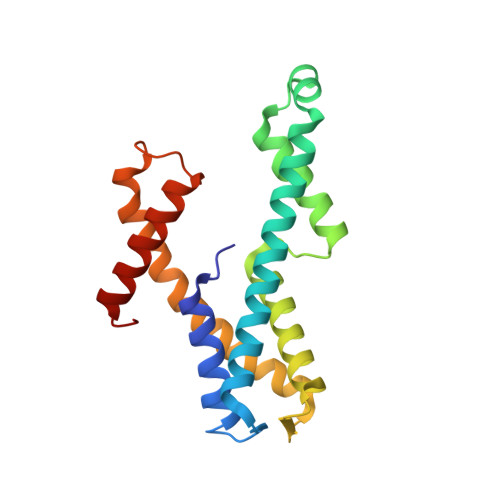
Crystal structure of domain of unknown function 507 (DUF507) reveals a new protein fold.
McKay, C.E., Cheng, J., Tanner, J.J.(2023) Sci Rep 13: 13496-13496
- PubMed: 37596303
- DOI: https://doi.org/10.1038/s41598-023-40558-y
- Primary Citation of Related Structures:
8T8K, 8T8L - PubMed Abstract:
The crystal structure of the domain of unknown function family 507 protein from Aquifex aeolicus is reported (AaDUF507, UniProt O67633, 183 residues). The structure was determined in two space groups (C222 1 and P3 2 21) at 1.9 Å resolution. The phase problem was solved by molecular replacement using an AlphaFold model as the search model. AaDUF507 is a Y-shaped α-helical protein consisting of an anti-parallel 4-helix bundle base and two helical arms that extend 30-Å from the base. The two crystal structures differ by a 25° rigid body rotation of the C-terminal arm. The tertiary structure exhibits pseudo-twofold symmetry. The structural symmetry mirrors internal sequence similarity: residues 11-57 and 102-148 are 30% identical and 53% similar with an E-value of 0.002. In one of the structures, electron density for an unknown ligand, consistent with nicotinamide or similar molecule, may indicate a functional site. Docking calculations suggest potential ligand binding hot spots in the region between the helical arms. Structure-based query of the Protein Data Bank revealed no other protein with a similar tertiary structure, leading us to propose that AaDUF507 represents a new protein fold.
Organizational Affiliation:
Department of Biochemistry, University of Missouri, Columbia, MO, 65211, USA.