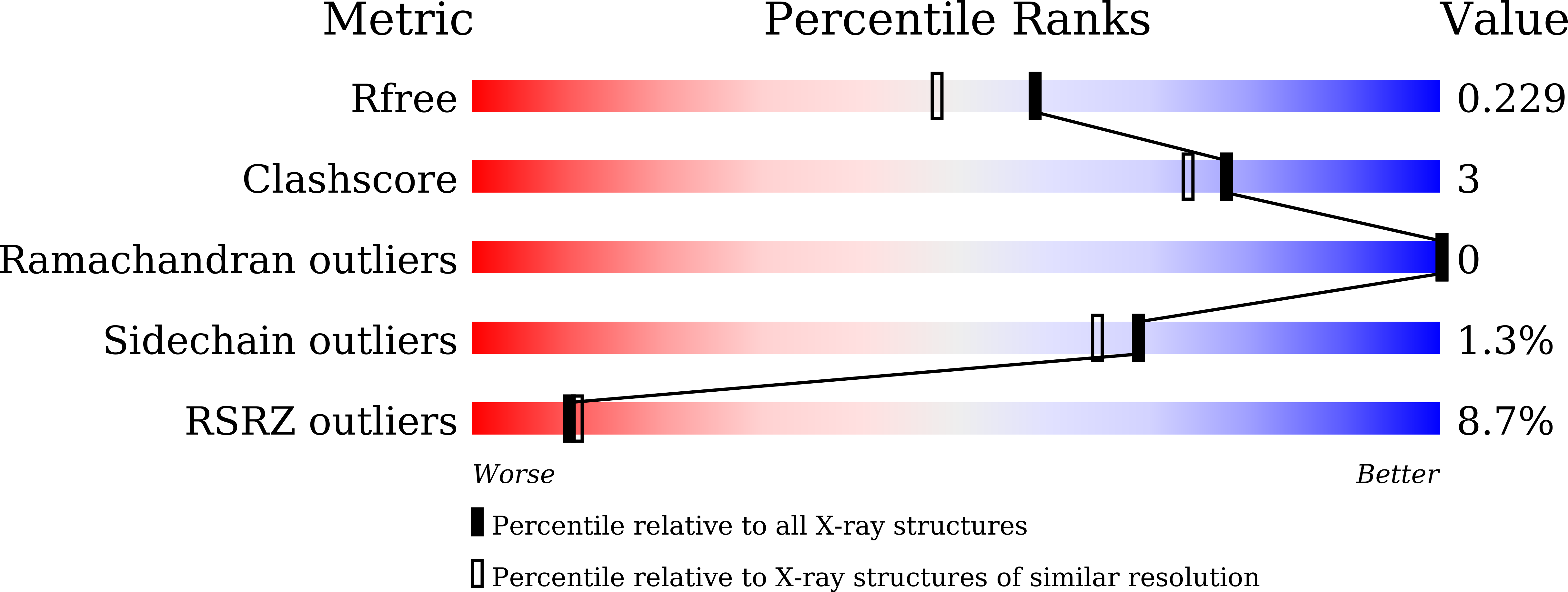
Structural mechanisms of calmodulin activation of Shigella effector OspC3 to ADP-riboxanate caspase-4/11 and block pyroptosis.
Hou, Y., Zeng, H., Li, Z., Feng, N., Meng, F., Xu, Y., Li, L., Shao, F., Ding, J.(2023) Nat Struct Mol Biol 30: 261-272
- PubMed: 36624349
- DOI: https://doi.org/10.1038/s41594-022-00888-3
- Primary Citation of Related Structures:
7WR0, 7WR1, 7WR2, 7WR3, 7WR4, 7WR5, 7WR6 - PubMed Abstract:
The caspase-4/11-GSDMD pyroptosis axis recognizes cytosolic lipopolysaccharide for antibacterial defenses. Shigella flexneri employs an OspC3 effector to block pyroptosis by catalyzing NAD + -dependent arginine ADP-riboxanation of caspase-4/11. Here, we identify Ca 2+ -free calmodulin (CaM) that binds and stimulates OspC3 ADP-riboxanase activity. Crystal structures of OspC3-CaM and OspC3-caspase-4 binary complexes reveal unique CaM binding to an OspC3 N-terminal domain featuring an ADP-ribosyltransferase-like fold and specific recognition of caspase-4 by an OspC3 ankryin repeat domain, respectively. CaM-OspC3-caspase-4 ternary complex structures show that NAD + binding reorganizes the catalytic pocket, in which D231 and D177 activate the substrate arginine for initial ADP-ribosylation and ribosyl 2'-OH in the ADP-ribosylated arginine, respectively, for subsequent deamination. We also determine structures of unmodified and OspC3-ADP-riboxanated caspase-4. Mechanisms derived from this series of structures covering the entire process of OspC3 action are supported by biochemical analyses in vitro and functional validation in S. flexneri-infected mice.
Organizational Affiliation:
National Laboratory of Biomacromolecules, CAS Center for Excellence in Biomacromolecules, Institute of Biophysics, Chinese Academy of Sciences, Beijing, China.