Discovery of a Highly Selective and H435R-Sensitive Thyroid Hormone Receptor beta Agonist.
Li, Q., Yao, B., Zhao, S., Lu, Z., Zhang, Y., Xiang, Q., Wu, X., Yu, H., Zhang, C., Li, J., Zhuang, X., Wu, D., Li, Y., Xu, Y.(2022) J Med Chem 65: 7193-7211
- PubMed: 35507418
- DOI: https://doi.org/10.1021/acs.jmedchem.2c00144
- Primary Citation of Related Structures:
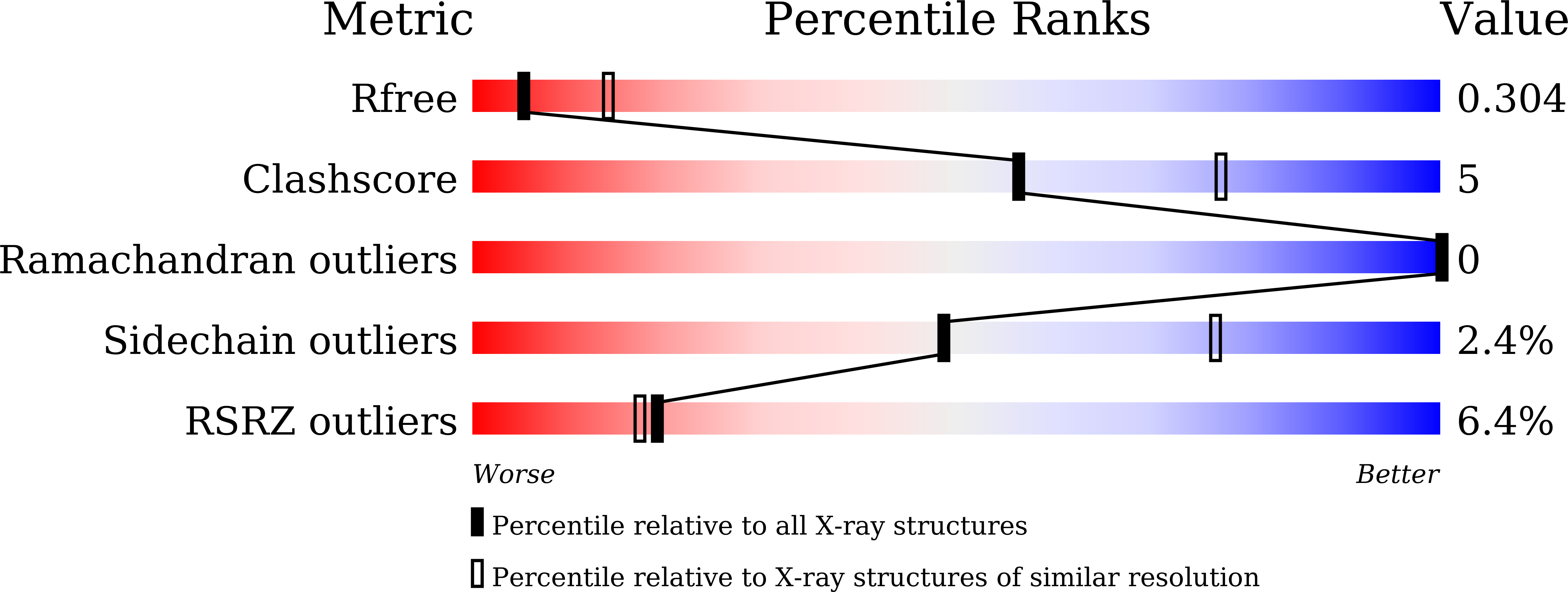
7WLX, 7WMG, 7WMH, 7WMJ, 7WML, 7WMN, 7WMO - PubMed Abstract:
The design and development of agonists selectively targeting thyroid hormone receptor β (TRβ) and TRβ mutants remain challenging tasks. In this study, we first adopted the strategy of breaking the "His-Phe switch" to solve two problems, simultaneously. A structure-based design approach was successfully utilized to obtain compound 16g , which is a potent TRβ agonist (EC 50 : 21.0 nM, 85.0% of the maximum efficacy of 1 ) with outstanding selectivity for TRβ over TRα and also effectively activates the TRβ H435R mutant. Then, we developed a highly efficient synthetic method for 16g . Our serials of cocrystal structures revealed detailed structural mechanisms in overcoming subtype selectivity and rescuing the H435R mutation. 16g also showed excellent lipid metabolism, safety, metabolic stability, and pharmacokinetic properties. Collectively, 16g is a well-characterized selective and mutation-sensitive TRβ agonist for further investigating its function in treating dyslipidemia, nonalcoholic steatohepatitis (NASH), and resistance to thyroid hormone (RTH).
Organizational Affiliation:
Center for Chemical Biology and Drug Discovery, Guangzhou Institutes of Biomedicine and Health, Chinese Academy of Sciences, Guangzhou 510530, China.