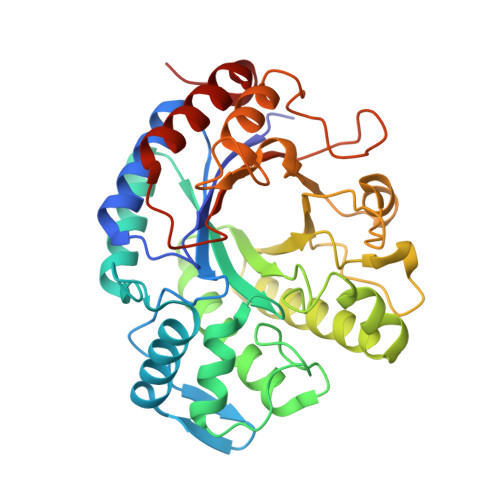
Mechanism of cooperative N-glycan processing by the multi-modular endoglycosidase EndoE.
Garcia-Alija, M., Du, J.J., Ordonez, I., Diz-Vallenilla, A., Moraleda-Montoya, A., Sultana, N., Huynh, C.G., Li, C., Donahue, T.C., Wang, L.X., Trastoy, B., Sundberg, E.J., Guerin, M.E.(2022) Nat Commun 13: 1137-1137
- PubMed: 35241669
- DOI: https://doi.org/10.1038/s41467-022-28722-w
- Primary Citation of Related Structures:
7PUJ, 7PUK, 7PUL - PubMed Abstract:
Bacteria produce a remarkably diverse range of glycoside hydrolases to metabolize glycans from the environment as a primary source of nutrients, and to promote the colonization and infection of a host. Here we focus on EndoE, a multi-modular glycoside hydrolase secreted by Enterococcus faecalis, one of the leading causes of healthcare-associated infections. We provide X-ray crystal structures of EndoE, which show an architecture composed of four domains, including GH18 and GH20 glycoside hydrolases connected by two consecutive three α-helical bundles. We determine that the GH20 domain is an exo-β-1,2-N-acetylglucosaminidase, whereas the GH18 domain is an endo-β-1,4-N-acetylglucosaminidase that exclusively processes the central core of complex-type or high-mannose-type N-glycans. Both glycoside hydrolase domains act in a concerted manner to process diverse N-glycans on glycoproteins, including therapeutic IgG antibodies. EndoE combines two enzyme domains with distinct functions and glycan specificities to play a dual role in glycan metabolism and immune evasion.
Organizational Affiliation:
Structural Glycobiology Laboratory, Biocruces Bizkaia Health Research Institute, Cruces University Hospital, 48903, Barakaldo, Bizkaia, Spain.