The acyl chains of phosphoinositide PIP3 alter the structure and function of nuclear receptor steroidogenic factor-1.
Bryant, J.M., Malabanan, M.M., Vanderloop, B.H., Nichols, C.M., Haratipour, Z., Poon, K.T., Sherrod, S.D., McLean, J.A., Blind, R.D.(2021) J Lipid Res 62: 100081-100081
- PubMed: 33933440
- DOI: https://doi.org/10.1016/j.jlr.2021.100081
- Primary Citation of Related Structures:
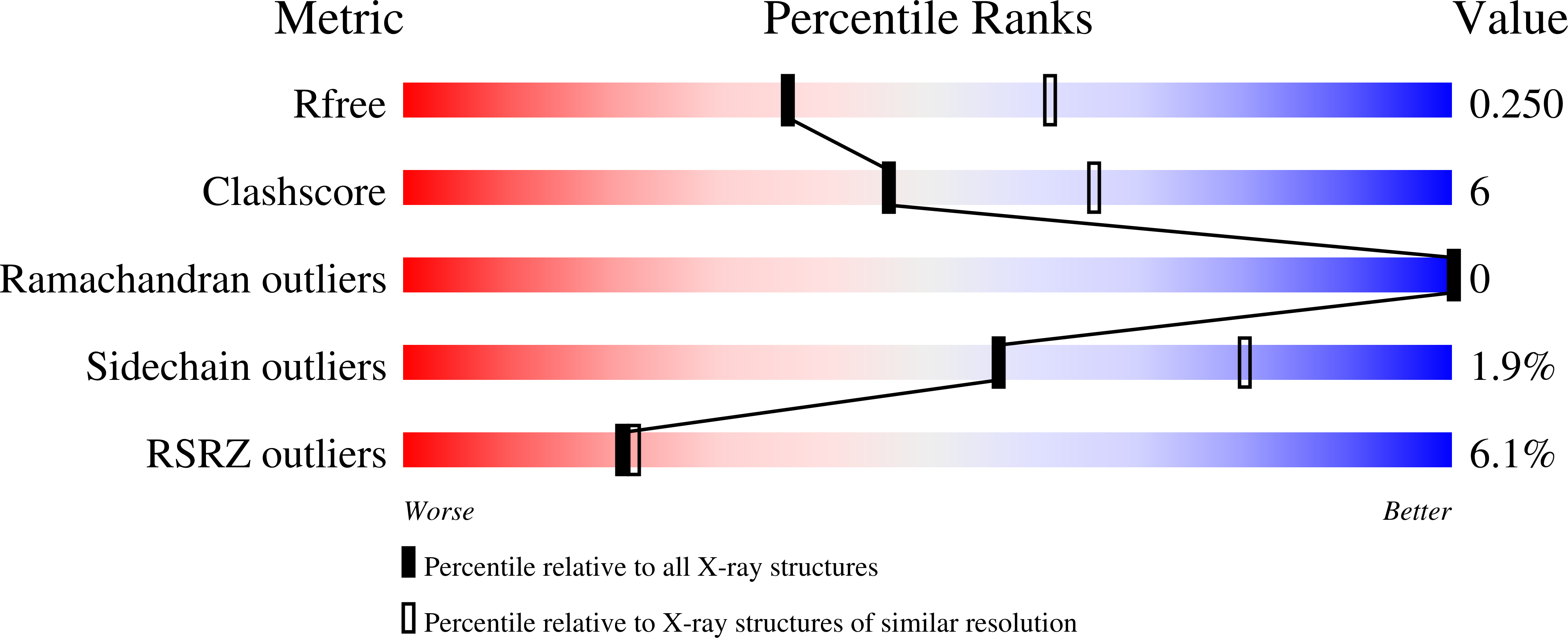
7KHT - PubMed Abstract:
Nuclear receptors are transcription factors that bind lipids, an event that induces a structural conformation of the receptor that favors interaction with transcriptional coactivators. The nuclear receptor steroidogenic factor-1 (SF-1, NR5A1) binds the signaling phosphoinositides PI(4,5)P2 (PIP2) and PI(3,4,5)P3 (PIP3), and our previous crystal structures showed how the phosphoinositide headgroups regulate SF-1 function. However, what role the acyl chains play in regulating SF-1 structure remains unaddressed. Here, we used X-ray crystallography with in vitro binding and functional assays to examine how the acyl chains of PIP3 regulate human SF-1 ligand-binding domain structure and function. Altering acyl chain length and unsaturation regulates apparent binding of all tested phosphoinositides to SF-1. Mass spectrometry-based lipidomics data suggest C16 and C18 phospholipids preferentially associate with SF-1 expressed ectopically in bacteria. We then solved the 2.5 Å crystal structure of SF-1 bound to dioleoyl PIP3(18:1/18:1) to compare it with a matched structure of SF-1 bound to dipalmitoyl PIP3(16:0/16:0). The dioleoyl-bound structure was severely disordered in a specific SF-1 region associated with pathogenic human polymorphisms and within the coactivator-binding region critical for SF-1 function while inducing increased sensitivity to protease digestion in solution. Validating these structural observations, in vitro functional studies showed dioleoyl PIP3 induced 6-fold poorer affinity of a peroxisome proliferator-activated receptor gamma coactivator 1-alpha coactivator peptide for SF-1 compared with dipalmitoyl PIP3. Together, these data suggest the chemical nature of the phosphoinositide acyl chains controls the ordered state of specific, clinically important structural regions in SF-1, regulating SF-1 function in vitro.
Organizational Affiliation:
Department of Pharmacology, Vanderbilt University School of Medicine, Nashville, TN, USA.