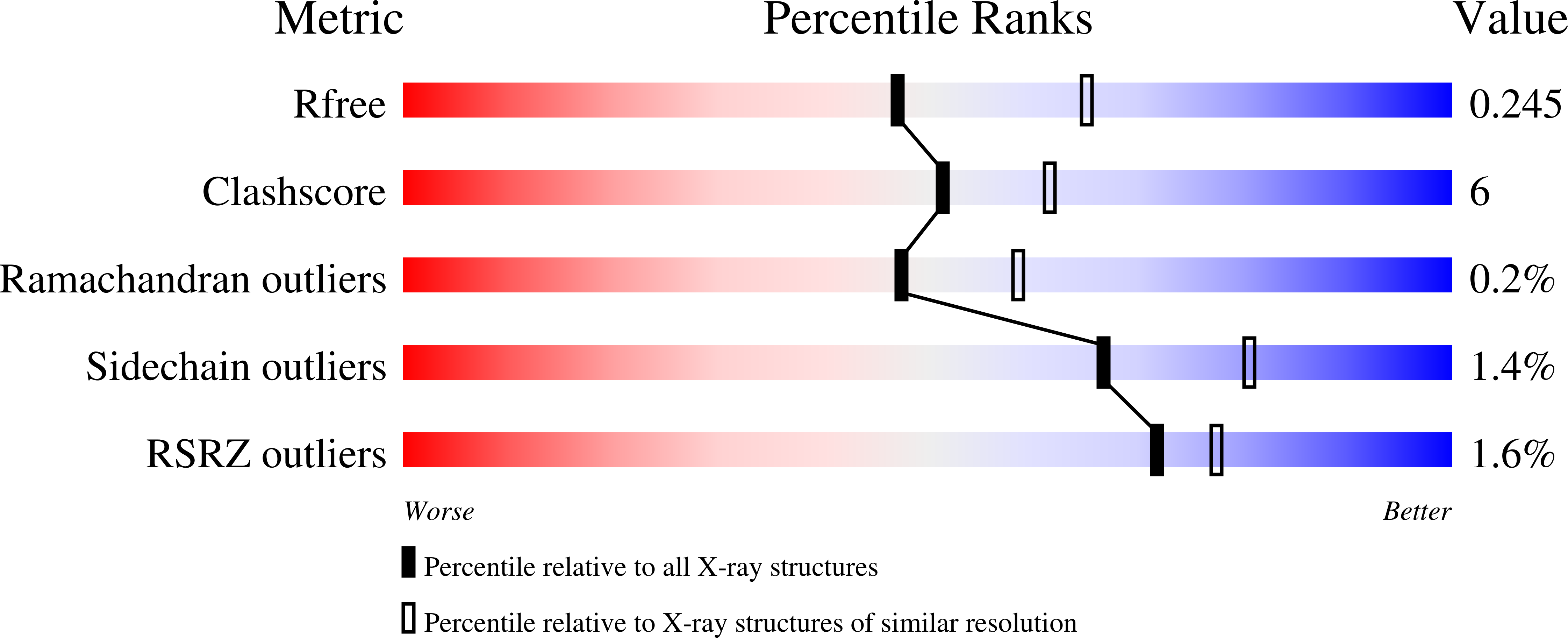
LanCLs add glutathione to dehydroamino acids generated at phosphorylated sites in the proteome.
Lai, K.Y., Galan, S.R.G., Zeng, Y., Zhou, T.H., He, C., Raj, R., Riedl, J., Liu, S., Chooi, K.P., Garg, N., Zeng, M., Jones, L.H., Hutchings, G.J., Mohammed, S., Nair, S.K., Chen, J., Davis, B.G., van der Donk, W.A.(2021) Cell 184: 2680-2695.e26
- PubMed: 33932340
- DOI: https://doi.org/10.1016/j.cell.2021.04.001
- Primary Citation of Related Structures:
6WQ1 - PubMed Abstract:
Enzyme-mediated damage repair or mitigation, while common for nucleic acids, is rare for proteins. Examples of protein damage are elimination of phosphorylated Ser/Thr to dehydroalanine/dehydrobutyrine (Dha/Dhb) in pathogenesis and aging. Bacterial LanC enzymes use Dha/Dhb to form carbon-sulfur linkages in antimicrobial peptides, but the functions of eukaryotic LanC-like (LanCL) counterparts are unknown. We show that LanCLs catalyze the addition of glutathione to Dha/Dhb in proteins, driving irreversible C-glutathionylation. Chemo-enzymatic methods were developed to site-selectively incorporate Dha/Dhb at phospho-regulated sites in kinases. In human MAPK-MEK1, such "elimination damage" generated aberrantly activated kinases, which were deactivated by LanCL-mediated C-glutathionylation. Surveys of endogenous proteins bearing damage from elimination (the eliminylome) also suggest it is a source of electrophilic reactivity. LanCLs thus remove these reactive electrophiles and their potentially dysregulatory effects from the proteome. As knockout of LanCL in mice can result in premature death, repair of this kind of protein damage appears important physiologically.
Organizational Affiliation:
Department of Biochemistry, University of Illinois at Urbana-Champaign, Urbana, IL 61801, USA.