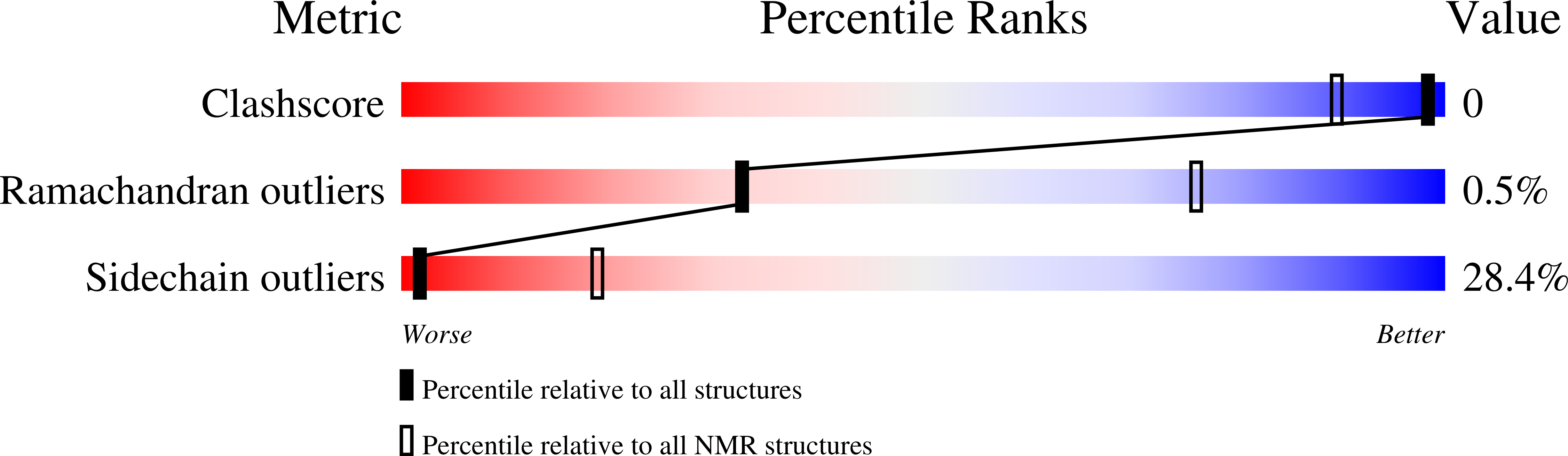Protein Allostery at Atomic Resolution.
Strotz, D., Orts, J., Kadavath, H., Friedmann, M., Ghosh, D., Olsson, S., Chi, C.N., Pokharna, A., Guntert, P., Vogeli, B., Riek, R.(2020) Angew Chem Int Ed Engl 59: 22132-22139
- PubMed: 32797659
- DOI: https://doi.org/10.1002/anie.202008734
- Primary Citation of Related Structures:
6SVC, 6SVE, 6SVH - PubMed Abstract:
Protein allostery is a phenomenon involving the long range coupling between two distal sites in a protein. In order to elucidate allostery at atomic resoluion on the ligand-binding WW domain of the enzyme Pin1, multistate structures were calculated from exact nuclear Overhauser effect (eNOE). In its free form, the protein undergoes a microsecond exchange between two states, one of which is predisposed to interact with its parent catalytic domain. In presence of the positive allosteric ligand, the equilibrium between the two states is shifted towards domain-domain interaction, suggesting a population shift model. In contrast, the allostery-suppressing ligand decouples the side-chain arrangement at the inter-domain interface thereby reducing the inter-domain interaction. As such, this mechanism is an example of dynamic allostery. The presented distinct modes of action highlight the power of the interplay between dynamics and function in the biological activity of proteins.
Organizational Affiliation:
Laboratory of Physical Chemistry, Swiss Federal Institute of Technology, ETH-Hönggerberg, 8093, Zürich, Switzerland.