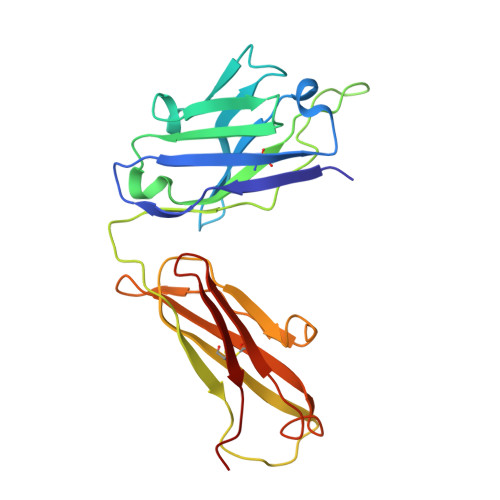
Crystal Structure of the Fab Fragment of an Anti-ofloxacin Antibody and Exploration of Its Specific Binding.
He, K., Du, X., Sheng, W., Zhou, X., Wang, J., Wang, S.(2016) J Agric Food Chem 64: 2627-2634
- PubMed: 26963935
- DOI: https://doi.org/10.1021/acs.jafc.5b05882
- Primary Citation of Related Structures:
4Z0B - PubMed Abstract:
The limited knowledge on the mechanism of interactions between small contaminants and the corresponding antibodies greatly inhibits the development of enzyme-linked immunosorbent assay methods. In this study, the crystal structure of a Fab fragment specific for ofloxacin was obtained. On the basis of the crystal characteristics, the modeling of the interactions between ofloxacin and the Fab revealed that TYR31 and HIS99 of the heavy chain and MET20 and GLN79 of the light chain formed a hydrophobic region and that SER52 and ALA97 of the heavy chain and TYR35 of the light chain formed a salt bridge and two hydrogen bonds for specific binding. The key roles of SER52 and ALA97 were further confirmed by site-directed mutation. A specificity analysis using 14 ofloxacin analogues indicates that the length of the bond formed between the piperazine ring and the antibody plays key roles in specific recognition. This work helps to clarify the mechanisms through which antibodies recognize small molecules and improve immune detection methods.
Organizational Affiliation:
Key Laboratory of Food Nutrition and Safety, Ministry of Education of China, Tianjin University of Science and Technology , Tianjin 300457, People's Republic of China.