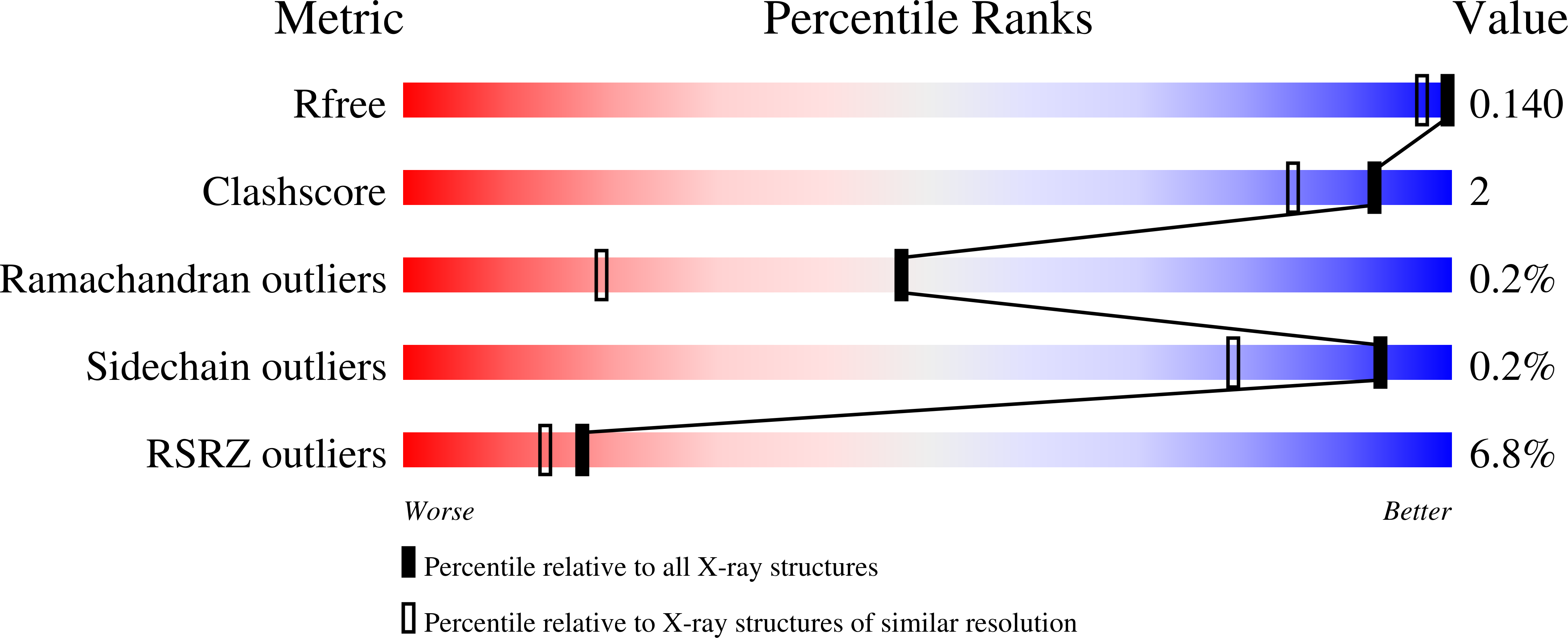
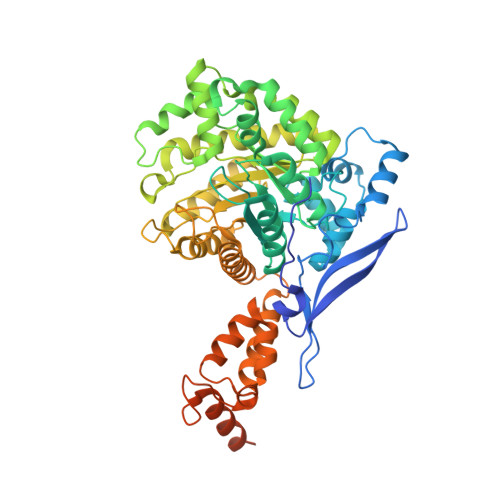
Non-canonical active site architecture of the radical SAM thiamin pyrimidine synthase.
Fenwick, M.K., Mehta, A.P., Zhang, Y., Abdelwahed, S.H., Begley, T.P., Ealick, S.E.(null) Nat Commun 6: 6480-6480
- PubMed: 25813242
- DOI: https://doi.org/10.1038/ncomms7480
- Primary Citation of Related Structures:
4S25, 4S26, 4S27, 4S28, 4S29, 4S2A - PubMed Abstract:
Radical S-adenosylmethionine (SAM) enzymes use a [4Fe-4S] cluster to generate a 5'-deoxyadenosyl radical. Canonical radical SAM enzymes are characterized by a β-barrel-like fold and SAM anchors to the differentiated iron of the cluster, which is located near the amino terminus and within the β-barrel, through its amino and carboxylate groups. Here we show that ThiC, the thiamin pyrimidine synthase in plants and bacteria, contains a tethered cluster-binding domain at its carboxy terminus that moves in and out of the active site during catalysis. In contrast to canonical radical SAM enzymes, we predict that SAM anchors to an additional active site metal through its amino and carboxylate groups. Superimposition of the catalytic domains of ThiC and glutamate mutase shows that these two enzymes share similar active site architectures, thus providing strong evidence for an evolutionary link between the radical SAM and adenosylcobalamin-dependent enzyme superfamilies.
Organizational Affiliation:
Department of Chemistry and Chemical Biology, Cornell University, 120 Baker Lab, Ithaca, New York 14853, USA.