Crystal Structure of Glucocerebrosidase Containing the N370S mutation: Implication on Chaperon Therapy
Wei, R.R., Boucher, S., Hughes, H., Guziewica, N., Vanpatten, S., Pan, C.Q., Edmunds, T.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Glucocerebrosidase | 497 | Homo sapiens | Mutation(s): 1 Gene Names: GBA, GC, GLUC EC: 3.2.1.45 (PDB Primary Data), 3.2.1.46 (UniProt), 2.4.1 (UniProt), 3.2.1 (UniProt) | 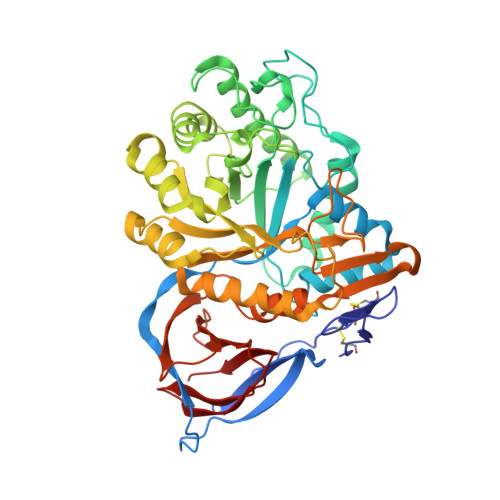 | |
UniProt & NIH Common Fund Data Resources | |||||
Find proteins for P04062 (Homo sapiens) Explore P04062 Go to UniProtKB: P04062 | |||||
PHAROS: P04062 GTEx: ENSG00000177628 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P04062 | ||||
Glycosylation | |||||
| Glycosylation Sites: 2 | Go to GlyGen: P04062-1 | ||||
Sequence AnnotationsExpand | |||||
| |||||
| Ligands 4 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| NAG Query on NAG | K [auth A], O [auth B] | 2-acetamido-2-deoxy-beta-D-glucopyranose C8 H15 N O6 OVRNDRQMDRJTHS-FMDGEEDCSA-N |  | ||
| NDG Query on NDG | D [auth A] | 2-acetamido-2-deoxy-alpha-D-glucopyranose C8 H15 N O6 OVRNDRQMDRJTHS-PVFLNQBWSA-N |  | ||
| SO4 Query on SO4 | E [auth A] F [auth A] G [auth A] H [auth A] I [auth A] | SULFATE ION O4 S QAOWNCQODCNURD-UHFFFAOYSA-L |  | ||
| GOL Query on GOL | J [auth A], P [auth B] | GLYCEROL C3 H8 O3 PEDCQBHIVMGVHV-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 107.83 | α = 90 |
| b = 285.92 | β = 90 |
| c = 92 | γ = 90 |
| Software Name | Purpose |
|---|---|
| HKL-2000 | data collection |
| MOLREP | phasing |
| REFMAC | refinement |
| MOSFLM | data reduction |
| SCALA | data scaling |