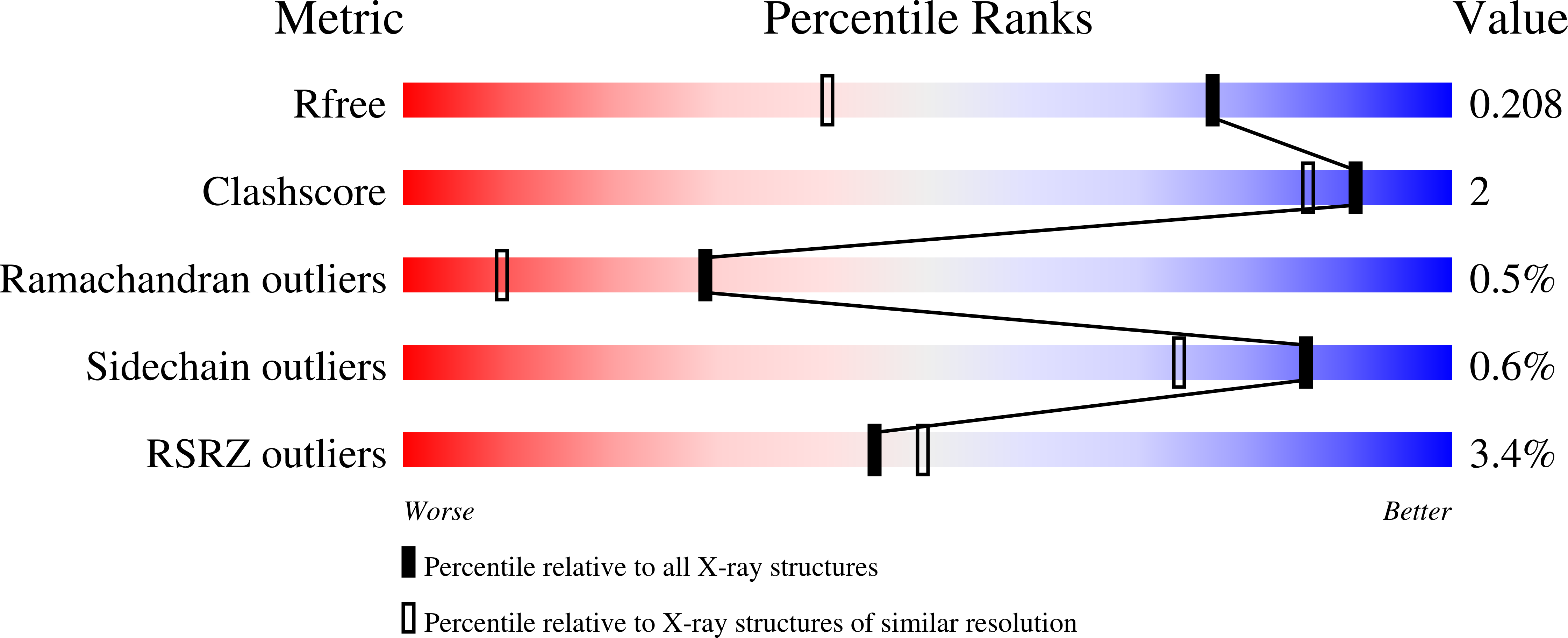
Crystal Structure and Enzymatic Properties of a Bacterial Family 19 Chitinase Reveal Differences with Plant Enzymes
Hoell, I.A., Dalhus, B., Heggset, E.B., Aspmo, S.I., Eijsink, V.G.H.(2006) FEBS J 273: 4889
- PubMed: 17010167
- DOI: https://doi.org/10.1111/j.1742-4658.2006.05487.x
- Primary Citation of Related Structures:
2CJL - PubMed Abstract:
We describe the cloning, overexpression, purification, characterization and crystal structure of chitinase G, a single-domain family 19 chitinase from the Gram-positive bacterium Streptomyces coelicolor A3(2). Although chitinase G was not capable of releasing 4-methylumbelliferyl from artificial chitooligosaccharide substrates, it was capable of degrading longer chitooligosaccharides at rates similar to those observed for other chitinases. The enzyme was also capable of degrading a colored colloidal chitin substrate (carboxymethyl-chitin-remazol-brilliant violet) and a small, presumably amorphous, subfraction of alpha-chitin and beta-chitin, but was not capable of degrading crystalline chitin completely. The crystal structures of chitinase G and a related Streptomyces chitinase, chitinase C [Kezuka Y, Ohishi M, Itoh Y, Watanabe J, Mitsutomi M, Watanabe T & Nonaka T (2006) J Mol Biol358, 472-484], showed that these bacterial family 19 chitinases lack several loops that extend the substrate-binding grooves in family 19 chitinases from plants. In accordance with these structural features, detailed analysis of the degradation of chitooligosaccharides by chitinase G showed that the enzyme has only four subsites (- 2 to + 2), as opposed to six (- 3 to + 3) for plant enzymes. The most prominent structural difference leading to reduced size of the substrate-binding groove is the deletion of a 13-residue loop between the two putatively catalytic glutamates. The importance of these two residues for catalysis was confirmed by a site-directed mutagenesis study.
Organizational Affiliation:
Department of Chemistry, Biotechnology and Food Science, Norwegian University of Life Sciences, 1432 As, Norway.