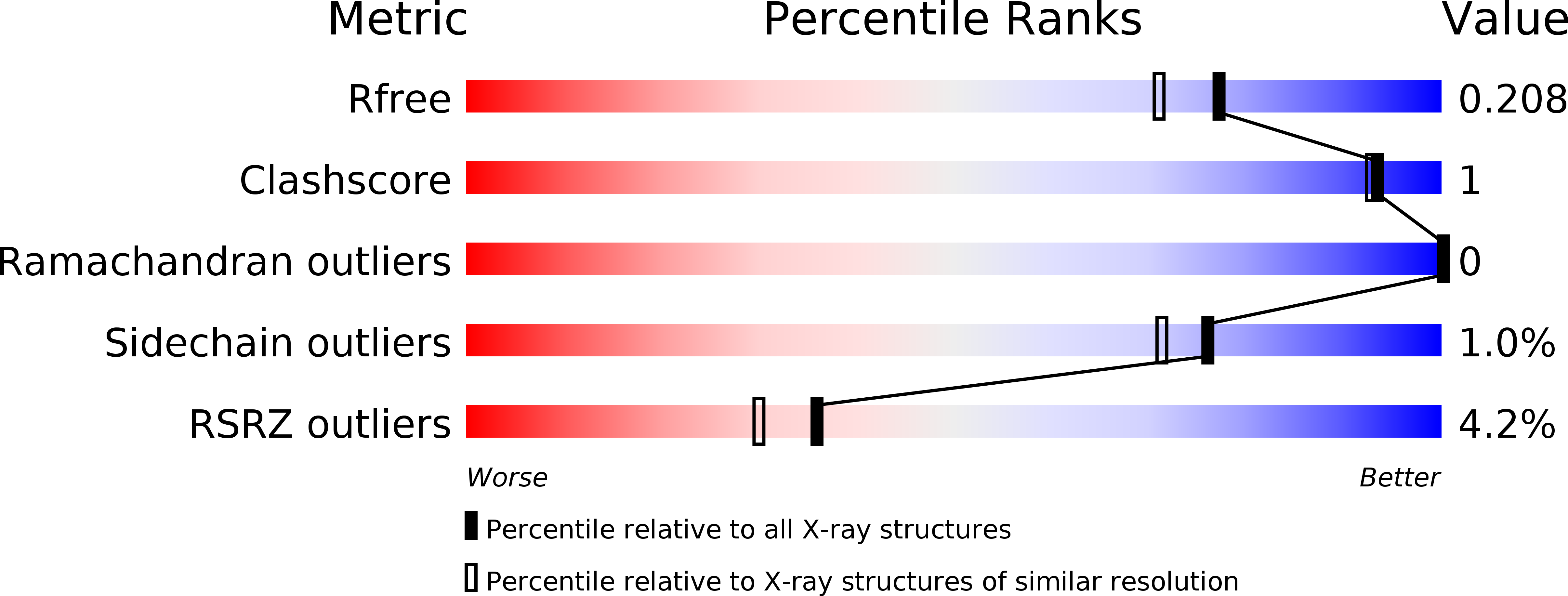
Enantiomer-Specific Binding of Ruthenium(II) Molecular Wires by the Amine Oxidase of Arthrobacter Globiformis.
Langley, D.B., Brown, D.E., Cheruzel, L.E., Contakes, S.M., Duff, A.P., Hilmer, K.M., Dooley, D.M., Gray, H.B., Guss, J.M., Freeman, H.C.(2008) J Am Chem Soc 130: 8069
- PubMed: 18507382
- DOI: https://doi.org/10.1021/ja801289f
- Primary Citation of Related Structures:
2CFD, 2CFG, 2CFK, 2CFL, 2CFW, 2CG0, 2CG1 - PubMed Abstract:
The copper amine oxidase from Arthrobacter globiformis (AGAO) is reversibly inhibited by molecular wires comprising a Ru(II) complex head group and an aromatic tail group joined by an alkane linker. The crystal structures of a series of Ru(II)-wire-AGAO complexes differing with respect to the length of the alkane linker have been determined. All wires lie in the AGAO active-site channel, with their aromatic tail group in contact with the trihydroxyphenylalanine quinone (TPQ) cofactor of the enzyme. The TPQ cofactor is consistently in its active ("off-Cu") conformation, and the side chain of the so-called "gate" residue Tyr296 is consistently in the "gate-open" conformation. Among the wires tested, the most stable complex is produced when the wire has a -(CH2)4- linker. In this complex, the Ru(II)(phen)(bpy)2 head group is level with the protein molecular surface. Crystal structures of AGAO in complex with optically pure forms of the C4 wire show that the linker and head group in the two enantiomers occupy slightly different positions in the active-site channel. Both the Lambda and Delta isomers are effective competitive inhibitors of amine oxidation. Remarkably, inhibition by the C4 wire shows a high degree of selectivity for AGAO in comparison with other copper-containing amine oxidases.
Organizational Affiliation:
School of Molecular and Microbial Biosciences, University of Sydney, NSW 2006, Australia.